Mutational Signatures (v3.6 - May 2026)
CN17 · GRCh37 · COSMIC v104
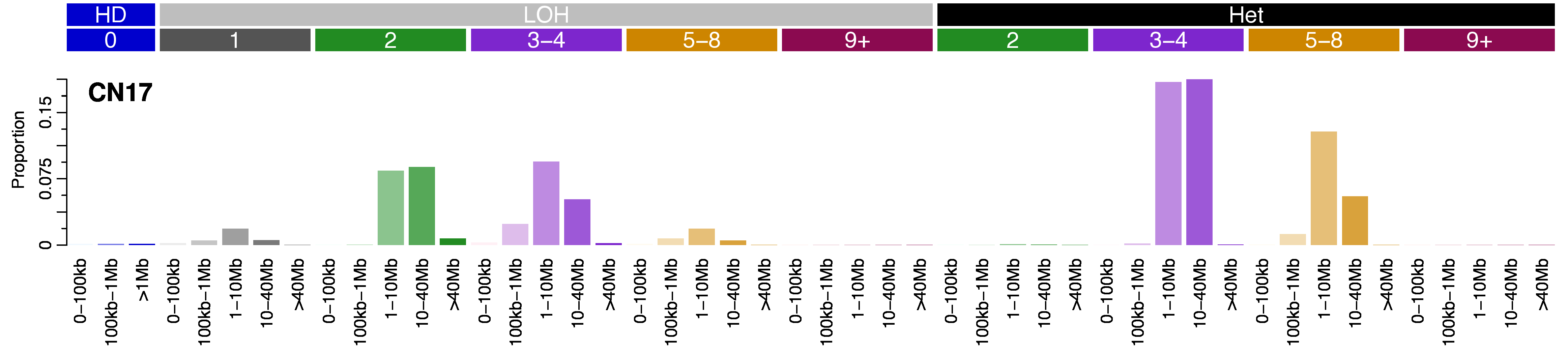
Mutational profile
Copy number signatures are defined by a 48 context copy number classification scheme. The scheme incorporates loss-of-heterozygosity status, total copy number state, and segment length to categorise segments from allele-specific copy number profiles.
The scheme corresponds to three heterozygosity states:
- heterozygous segments with copy number of [A>0, B>0] (numbers reflect the counts for major allele A and minor allele B)
- segments with LOH with copy number of [A>0, B=0]
- segments with homozygous deletions [A=0, B=0]
Segments are further subclassified into 6 classes based on the sum of major and minor allele (total copy number) and chosen for biological relevance:
- total copy number (TCN)=0 (homozygous deletion)
- TCN=1 (deletion leading to LOH)
- TCN=2 (wild type, including copy-neutral LOH)
- TCN=3 or 4 (minor gain)
- TCN=5 to 8 (moderate gain)
- TCN>=9 (high-level amplification).
Each of the heterozygous and LOH total copy number states are then organised into five classes based on the size of their segments: 0 – 100kb, 100kb – 1Mb, 1Mb – 10Mb, 10Mb – 40Mb, and >40Mb (the largest category for homozygous deletions was restricted to >1Mb) in order to capture focal, large scale, and chromosomal-scale copy number changes. This gives a total of 48 copy number context to categorise segments into (3x homozygous deletion categories, 5*5=25x LOH categories, 4*5=20 heterozygous categories).
Proposed aetiology
CN17 is a signature of homologous recombination deficiency (HRD). Evidence for this comes from positive associations with mono- or bi-allelic losses of BRCA1, BRCA2, PALB2, FBXW7 and CDK12, a positive association with HR pathway abrogation more generally, and the strongest positive association with scarHRD scores (a score of genomic scars of homologous recombination deficiency, see Sztupinszki et al., 2018). In addition, CN17 associates with promoter hypermethylation of BRCA1 in breast cancer, and with SBS3 (a single base substitution signature associates with homologous recombination deficiency), ID6 (an indel signature associated with homologous recombination deficiency), and ID8 (an indel signature associated with non homologous end joining, which is often preferred when homologous recombination is unavailable) in whole-genome-sequencing derived signature assignments. In addition, CN17 is positively associated with SBS2 and SBS13 (single base substitution signatures associates with APOBEC activity, and often observed near rearrangements/double strand breaks) in exome-derived signature assignments. CN17 is further positively associates with TP53 mutations in multiple cancers. CN17 is also associated with deletion and duplication rearrangement classes (see Hadi et al., 2020), similar to other CIN signatures, and indeed, CN17 is observed in a minority of high CIN simulations.
The tandem duplicator phenotype is often seen in samples that are jointly mutated for BRCA1 and TP53; CN17 is enriched in tandem duplicator phenotype samples (see Menghi et al., 2018), is positively associates with TP53 mutation in multiple cancer types,
In addition to HRD, CN17 is also associated with increased hypoxia levels (see Eustace et al., 2013 & Yang et al., 2018). Hypoxia has been shown to downregulate homologous recombination (see Bindra et al., 2004), making it possible that some of the attribution of CN17 is related to hypoxia as opposed to true HRD, however, associations with e.g. BRCA1 mutation status, suggest CN17 is at least partially a true signature of HRD.
Beyond associations directly relevant to HRD, CN17 is also associated with samples that are once-genome-doubled, with amplification of MYC in breast cancer, NKX2-1 in lung adenocarcinoma and AKT2 in uterine/endometrial carcinoma, and with an increased leucocyte fraction in breast cancer (see Thorsson et al., 2018).
CN17 is also associated with a poor prognosis for patients in adrenocortical carcinoma and uterine/endometrial carcinoma, and associated with patients with black ancestry over those with white ancestry.
Acceptance criteria
| Background | Identification study | First included in COSMIC | |
|---|---|---|---|
| Steele et al. 2022 Nature | v3.3 | ||
| Identification | NGS technique | Different variant callers | Multiple sequencing centres |
| SNP6, WES | No | No | |
| Technical validation | Validated in orthogonal techniques | Replicated in additional studies | Extended context enrichment |
| No | No | - | |
| Proposed aetiology | Mutational process | Support | |
| HR deficiency | Statistical association | ||
| Experimental validation | Experimental study | Species | |
| - | - | ||
Summary of the technical and experimental evidence available in the scientific literature regarding the validation of the mutational signature.
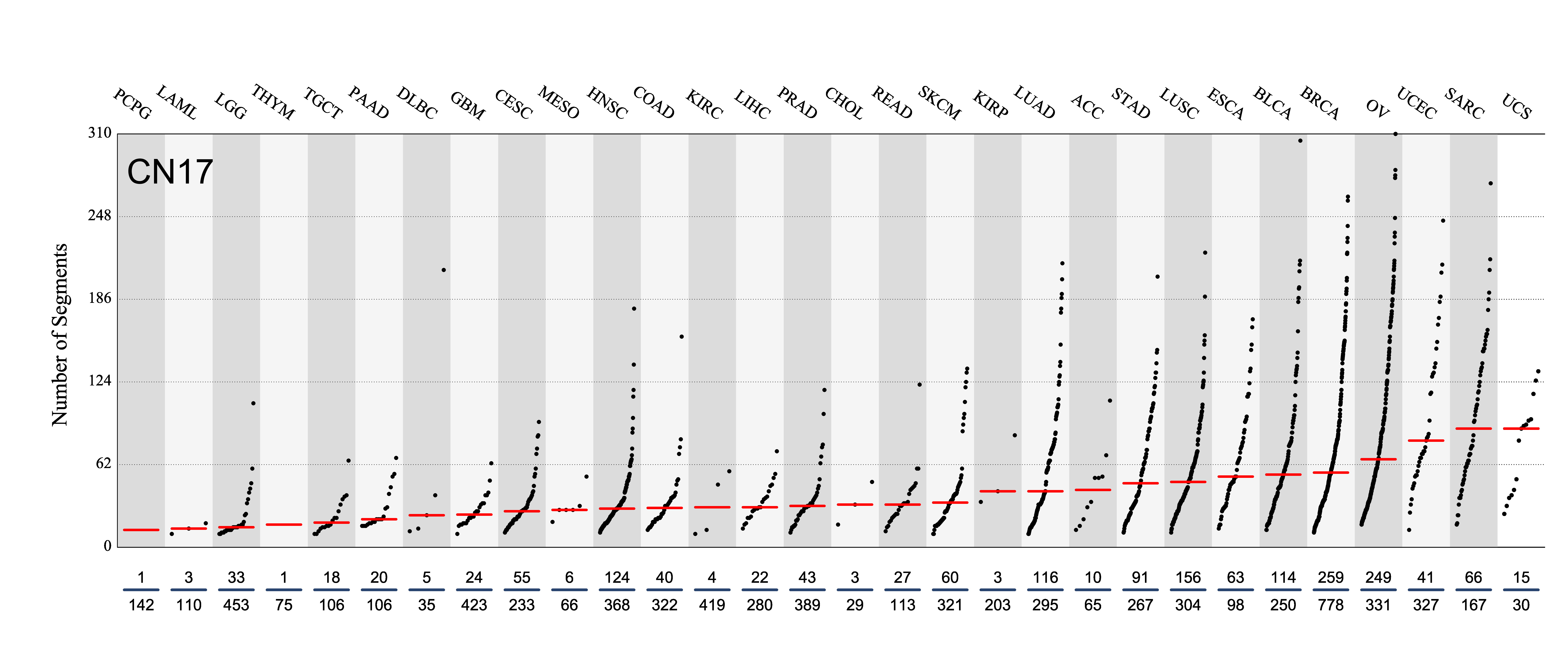
Tissue distribution
Number of segments attributed to the copy number signature across the cancer types in which it was found. Each dot represents an individual sample and only samples where the signature is found are shown.
The numbers below the dots for each cancer type indicate the number of high confidence tumours in which at least 10 mutations were attributed to the signature (above the blue horizontal line) and the total number of high confidence tumours analysed (below the blue horizontal line). Only high confidence data are displayed: samples with reconstruction accuracy >0.90. Note that due to the nature of copy number data, a sample with e.g. 22 counts of CN1 (a majority diploid unaltered signature, with large segment sizes) may span the whole genome, while a sample with e.g. 70 counts of CN8 (a highly segmented chromothripsis amplification signature, with small segment sizes) may span a small region of the genome.